|
While whole-genome and whole-exome sequencing experiments are exploited to investigate the entire genetic heritage of an individual, targeted re-sequencing applications have been introduced for those investigations where only small, user-defined portions of the genome need to be sequenced. NGS techniques generate high-throughput genomic data and specific analysis procedures (called bioinformatic pipelines) are currently developed to extract the information of interest from the extremely large amount of raw data generated as output from NGS experiments. Recent years have witnessed the advent of Next Generation Sequencing (NGS) technologies that have greatly improved sequencing capability, while dramatically decreasing the cost per sequenced base. In the last 30 years, Sanger sequencing has been the gold standard technique in molecular diagnostics. Our method shows how to manage one of these complex configurations, when two mutations occur. Although a consensus has been established in the elimination of primer sequences from aligned data (e.g., via primer sequence trimming or soft clipping), more complex configurations need to be managed in order to increase the retrieved information from available data. ConclusionsĪllele drop-out plays a crucial role in amplicon-based targeted re-sequencing and specific procedures in data analysis of amplicon data should be adopted. In this configuration, our proposed methodology outperforms Custom Amplicon workflow, being able to correctly identify two mutations in all the studied configurations. Results show that 14 % of identified variants is potentially affected by allele drop-out artifacts and that both the Custom Amplicon workflow and the procedure proposed here could correctly identify them.įurthermore, a more complex configuration in presence of two mutations was simulated in silico. Resultsįour gene panels, specific for Parkinson disease, for Intracerebral Hemorrhage Diseases (COL4A1 and COL4A2 genes) and for Familial Hemiplegic Migraine (CACNA1A and ATP1A2 genes) were designed.Ī total of 119 samples were re-sequenced with Illumina MiSeq sequencer and panel characterization in terms of coverage, number of variants found and allele drop-out potential impact has been carried out. This methodology has been compared to the Illumina Custom Amplicon workflow, available on Illumina MiSeq, on the analysis of data obtained with four newly designed TruSeq Custom Amplicon gene panels. Here, a procedure to manage such artifacts, based on a pipeline composed of two steps of alignment and variant calling, is proposed. Between these artifacts, allele drop-out, which is the preferential amplification of one allele, causes an artificial increase in homozygosity when heterozygous mutations fall on a primer pairing region. The reliability of such approaches rests on the high specificity and deep coverage, although sequencing artifacts attributable to PCR-like amplification can be encountered. Validating custom primers for sequencing on the iSeq 100 Sequencing System.Amplicon-based targeted resequencing is a commonly adopted solution for next-generation sequencing applications focused on specific genomic regions. Illumina cannot guarantee the performance or compatibility of custom primers. Refer to VP10 and VP14 Custom Primers for more information. Each primer is loaded into the library reservoir individually.ĭepending on your library preparation kit, you might be required to use Illumina primer mixes. The iSeq 100 allows the use of two custom read primers and two custom index primer combinations. The iSeq 100 Sequencing System support pages on the Illumina support center provides additional resources. To use custom primers with the iSeq 100 i1 Reagent v2 kits, update and install iSeq 100 Control Software v3.0 or later. Prepare and add the appropriate volume of each custom primer to the library reservoir of the cartridge after pre-run checks are completed. Select the custom primers in the control software during run setup. Set the iSeq 100 to manual run mode and enable the custom primers setting. 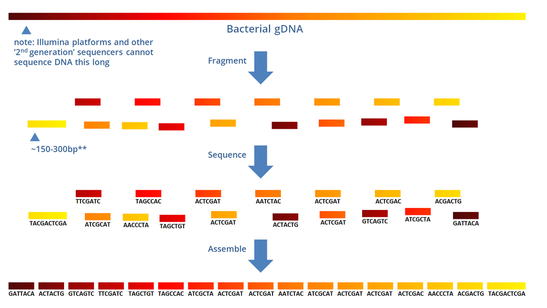
Using custom primers for a run on an Illumina iSeq 100 Sequencing System requires three additional steps at the run setup: Follow the run setup instructions described in the Run Settings.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
